-
Introduction
View the activity introduction for more information on the topic
-
Read aloud
Read the question aloud
-
Accessibility
Open the accessibility toolbar to change fonts and contrast, choose a different language, use a ruler and more
When a chemical reaction takes place, there are several changes that may occur. These visible changes are called the indicators of a chemical reaction. There are several indicators of reaction:
Colour Change

A change in colour is a sign of a chemical reaction. For example, frying an egg is a chemical reaction with a colour change - colourless to white.
Gas Given Off

During a chemical reaction, a gas can be produced. This can be seen by bubbles forming in the reaction or by a coloured gas being produced (such as smoke).
Solid Formed

If two solutions are mixed and a solid is formed, this is called a precipitate. A precipitate is an insoluble solid formed by reacting two liquids. For example, when milk goes very sour, a solid is formed.
Temperature Change

An energy change involves a change in temperature (either up or down). For example, the burning of a match involves a release of both heat and light.
In a chemical reaction, one or more new substances are always produced. So it is difficult to reverse a chemical reaction, as atoms have been rearranged.
So what is a physical change?
A physical change is where a substance just changes state. Physical changes are reversible - we always end up with the same substance we started with but sometimes in a different state.
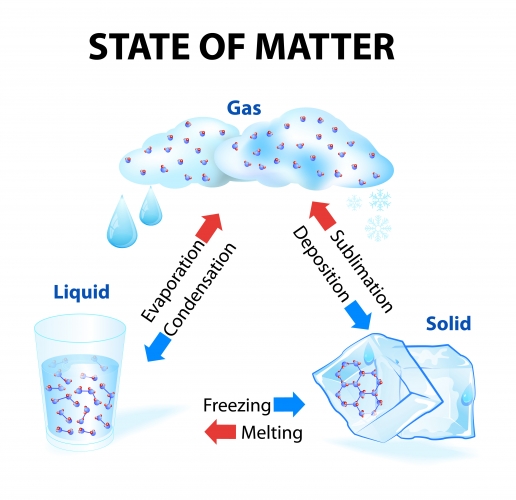
Let's have a go at some questions now.
Question
/ 10
Mr Jones
Do you want to skip questions and finish?
Hold up, it looks like there may be an issue with your spelling. Why not try it again?
You need to check your answer before you proceed. Are you sure you want to skip?

Teacher explanation