What happens to the ice in your drink on a hot day, and what happens to the water vapour in the air as it touches a cold window?

That's right, they change state. The particles in the ice and the water vapour are behaving differently as they are heated or cooled. Let's find out more.
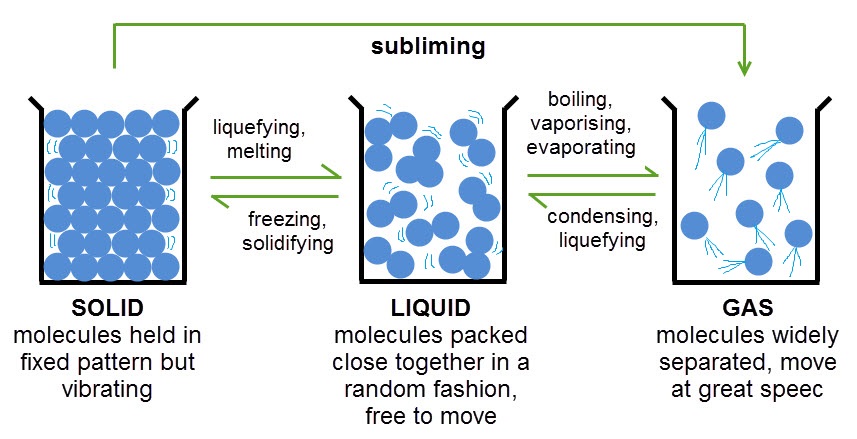
As you can see, when heated, particles in a substance gain energy. This means that they can move around more and when they gain enough energy, they can move further apart and eventually change state. The opposite happens when a substance is cooled. The particles lose energy and become closer together.
There are some important words in the diagram above that you'll need to remember about state changes. One that you may not have come across before is subliming. Sublimation happens when a solid is heated and changes directly into a gas, skipping the liquid state altogether. This is what happens to solid carbon dioxide, or 'dry ice' as it is known.
Got it? Let's try some questions on heating and cooling and the resulting changes of state.








