We know about the three different states of matter - solids, liquids and gases - but now we are going to think a bit more deeply about what the particles are doing in them when they change state, and why.
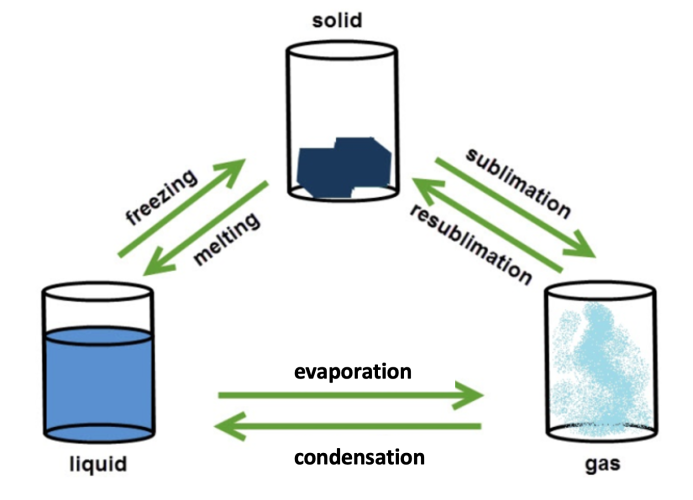
When heated, particles in a substance absorb energy from their surroundings. This means that they can move around more and much faster and when they gain enough energy, they can move further apart and eventually change state.
The opposite happens when a substance is cooled. The particles transfer some of their energy to the surroundings and become slower and closer together. You can see this in the diagram above - the particles in the gas are far apart and free to move independently of each other. As the gas cools to a liquid, the particles get closer together, and in a solid the particles are held together by strong forces of attraction. This is why particles in a solid can't move much, they can only vibrate on the spot.
There are some important words in the diagram above that you'll need to remember about state changes.
Freezing and condensing are the words we use to describe changes of state when energy is released.
Evaporating (or boiling), melting and subliming are the words we use to describe changes of state when energy is absorbed.
You've probably come across most of these before. One that you may not have come across before is subliming. Sublimation happens when a solid is heated and changes directly into a gas, skipping the liquid state altogether. This is what happens to solid carbon dioxide, or 'dry ice' as it is known.
Temperature determines whether a substance will be in the solid, liquid or gaseous state, and at what point it will change state.
All substances have a melting point - the temperature at which a solid turns into a liquid. At a temperature above the melting point, the substance will be a liquid, at a temperature below the melting point, the substance will be a solid. So you could also call it the freezing point!
Substances also have a boiling point - the temperature at which a liquid turns into a gas. At a temperature above the boiling point, a substance will be a gas. At a temperature below the boiling point, it will be a liquid.
Does that all make sense?

Hopefully! Now we shall take a look at some questions on changes of state.








