If you had a mixture of different liquids, how could you separate them?
Well, most substances have different boiling points - the temperature at which they turn from a liquid to a gas.
Fractional distillation is used to separate a mixture of liquids with different boiling points.
In the science laboratory, a fractionating column along with a condenser is used to allow the liquids to reach their boiling point then separate into the different groups or fractions. You can see how this apparatus is set up in the lab in the image below:
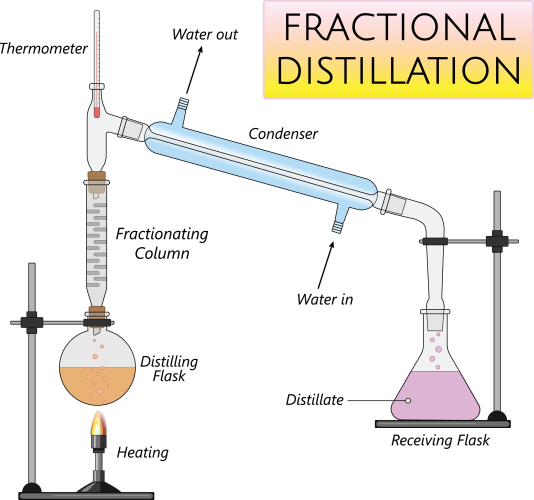
Crude oil is a mixture of different liquids. Fractional distillation can be used to separate crude oil into the many compounds that it is made up of, as they all have different boiling points. This is very important as crude oil contains some very useful compounds such as petrol and diesel. Fractional distillation of crude oil takes place on a large scale, in a huge fractionating column.
The products of fractional distillation of crude oil are shown in the diagram below:
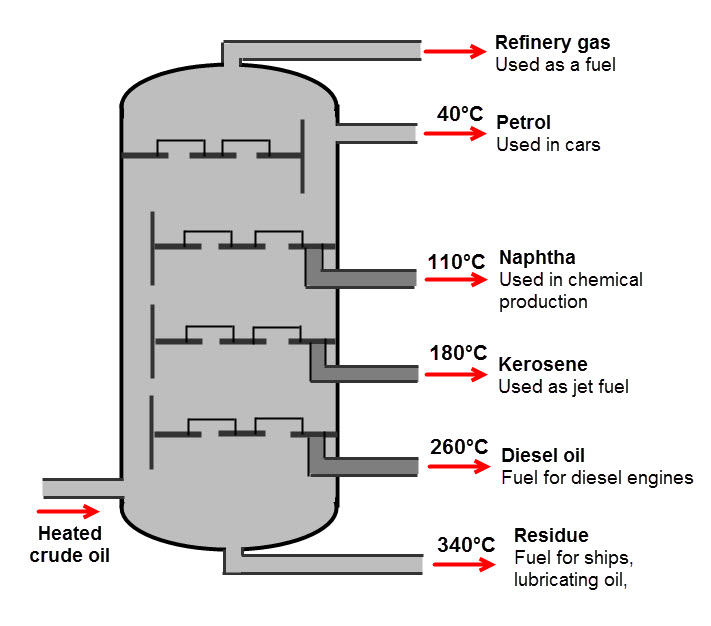
We can see that from crude oil there are lots of different, useful fractions produced. This is a reason why fractional distillation is so great!
Shall we try the questions on separating mixtures of liquids?








