Without enzymes, we could not function at all!
Every chemical reaction our body needs to survive, such as respiration, digestion and even thinking, all depend on fast reactions maintained by enzymes.
Both chemistry and biology are mad about enzymes, so let’s get going!
What are they?
Enzymes are actually a type of protein made by cells such as stomach cells.
Reminder: A protein or polypeptide is a folded polymer with a specific sequence of amino acids.
This arrangement of amino acids exists in a chain, which then folds and is held in place by chemical bonds, giving a unique 3D shape.
For enzymes, the final structure always includes an area designed for a specific molecule called an active site, and the shape is always specific to its function - for instance, an enzyme for insulin needs a different shape to an enzyme for starch.
What do they do?
They are biological catalysts!
A catalyst speeds up reactions without being used up.
They are specifically designed for certain substances meaning that every active site is a unique match to a certain ingredient ( we use the term 'reactants' in chemistry and 'substrates' in enzyme talk).
But remember - NO FIT = NO REACTION!
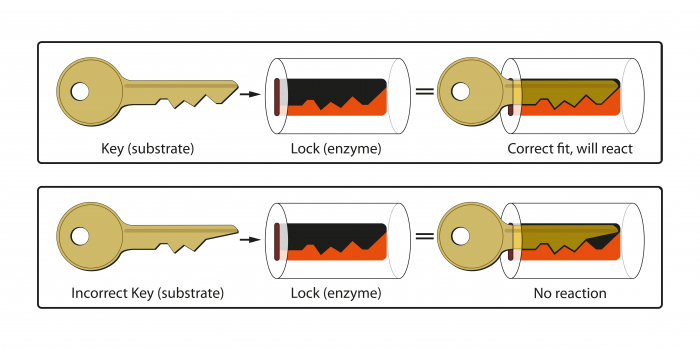
How do they work?
They use the lock and key hypothesis:
The correct enzyme needs to be present for the specific reaction, with its active site being the right fit for the starting substrate.
The substrate binds to the enzyme in the active site to make the enzyme-substrate complex.
From here, the enzyme has a different effect depending on the reaction, sometimes breaking down the substrate or even forming the desired product, but the enzyme always goes on to be reused.
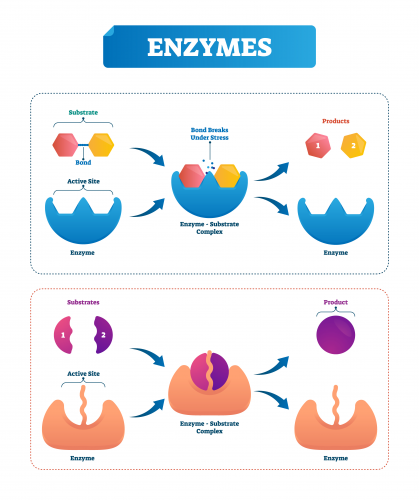
Factors for optimal enzyme activity
The factors to consider with enzymes include their optimal temperature and pH!
Temperature
There are three key points here:
Every enzyme has its optimum temperature, otherwise, their chemical bonds break and their active sites denature.
Changing the temperature will change the number of successful molecular collisions.
Too much heat can lead to denaturation. Denaturation means the active site starts to break down because too high a temperature is breaking the chemical bonds, ruining the active site and slowing the reaction.
Regarding molecular collisions, the enzyme and the substrate need kinetic energy to move around, so they have a better chance of hitting each other and reacting. But if the temperature dips too low, there is less movement and so less chance of successful collisions being made, slowing down the reaction.
However, it’s important to note that not every collision leads to a successful complex as sometimes the substrate goes into the active site but doesn’t react. Generally, though, optimum conditions mean more collisions and so successful collisions over time are more likely, maintaining a fast rate of reaction.
pH
This can also have a big impact on the active site shape, again because of the sensitivity of the chemical bonds between the amino acids in the folded 3D structure.
This leaves the active site vulnerable and if the pH makes a huge change outside the enzyme’s comfort zone, the active site will be denatured, and the substrate won’t be able to fit, which, you guessed it, slows the reaction right down!
Now it's time to have a go at some questions.







