Enzymes occur naturally in all organisms and are needed for many processes in the human body and in other organisms like plants. Enzymes are biological catalysts – they speed up the chemical reactions that take place inside all cells, but don't get used up in the process. Enzymes are protein molecules and are made up of amino acids. Most enzymes contain between 100 and 1,000 amino acids! These amino acids are joined together in a long chain, which is folded to produce a unique 3D structure. There are many different types of enzyme and each one catalyses a different reaction.
Enzymes can be found inside the cell, for example in the cytoplasm, mitochondria, chloroplast (in plants) or nucleus. Enzymes found here help with different processes like respiration, DNA replication, metabolism or photosynthesis.
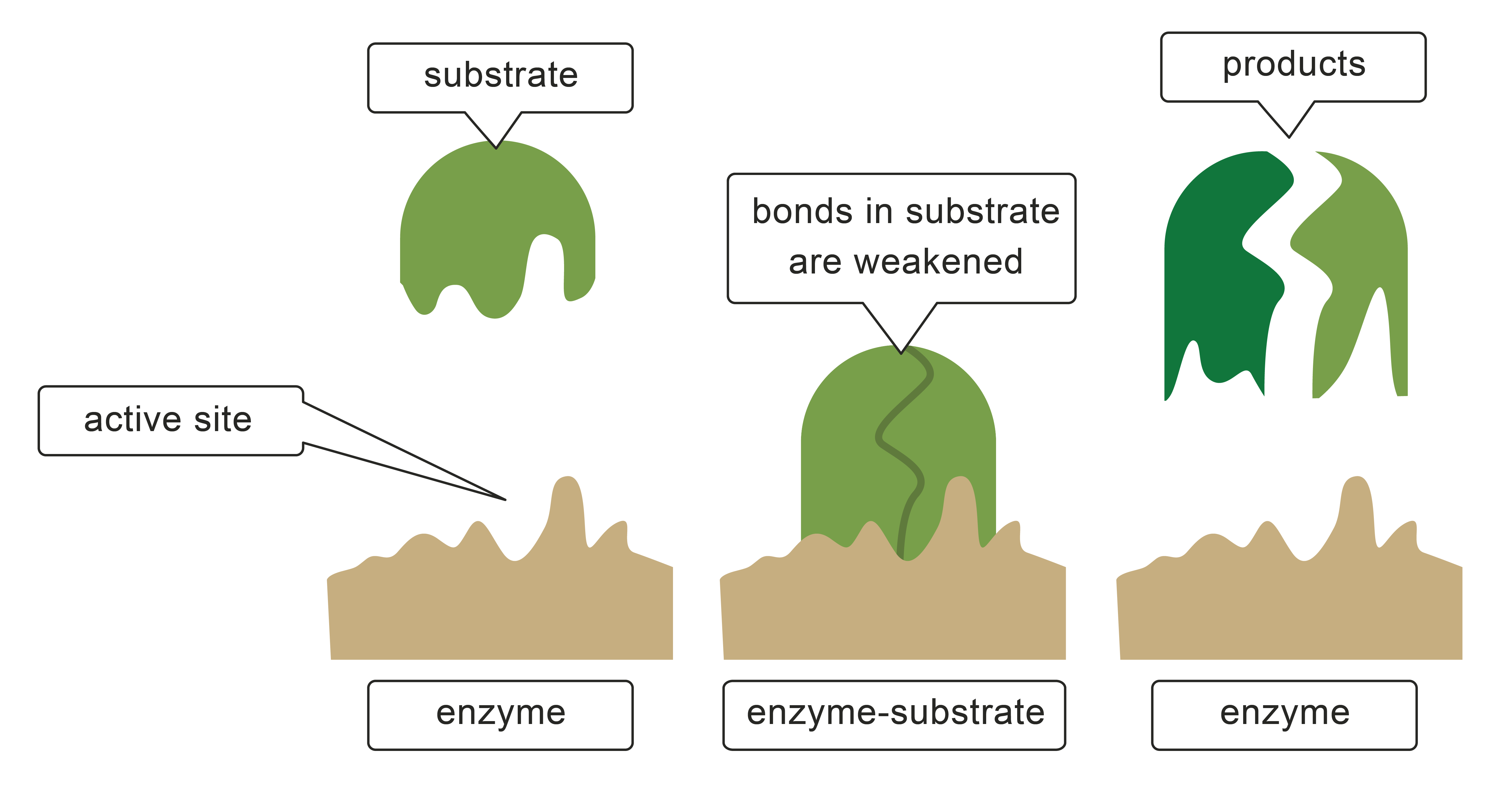
Enzymes have a specific shape that allows molecules to fit in called the active site. It's here that large substances are broken into smaller molecules. These smaller molecules might be used for different processes, for example, glucose can be used for respiration. Some smaller molecules will be used to build and make new products. Proteins can be broken down into amino acids which can be reassembled into new proteins.
The lock and key theory
The shape of the active site of an enzyme matches the shape of the substrate, a bit like a key is specific to the lock it opens, a substrate will only match a specific enzyme. This is called the lock and key theory.
Different factors can affect enzyme activity such as temperature, pH of the enzymes surroundings and the concentration of substrates.
At low temperatures the reaction between enzyme and substrate is slow. This is because, in order for an enzyme to catalyse a substrate, the substrate has to collide with enough energy with the enzymes active site. Low temperatures mean particles have less energy for collisions to occur. The reaction happens slower.
Higher temperatures increase the reaction between the enzyme and substrate. Substrates will have more energy allowing more successful collisions to occur between the enzyme and substrate, allowing the substrate to be catalysed at a faster rate.
However, too high temperatures can be problematic. Most enzymes have an optimum temperature, a temperature they work best at. At very high temperatures the active site of an enzyme denatures. This means the active site changes shape permanently and substrates can't fit into the active site anymore. These substrates won't be broken down. Body temperature for humans is 37°C. If temperatures reach above 37°C the enzymes in the body will start to denature. Different organisms have enzymes that work best at different temperatures.
Plants photosynthesise by using enzymes in their chloroplasts. Different types of plants grow best in different conditions; some plants grow best in very warm conditions while others can thrive in colder climates. This is why plants enzymes have a range of temperatures they work best in, unlike humans whose optimum temperature for enzymes is body temperature.
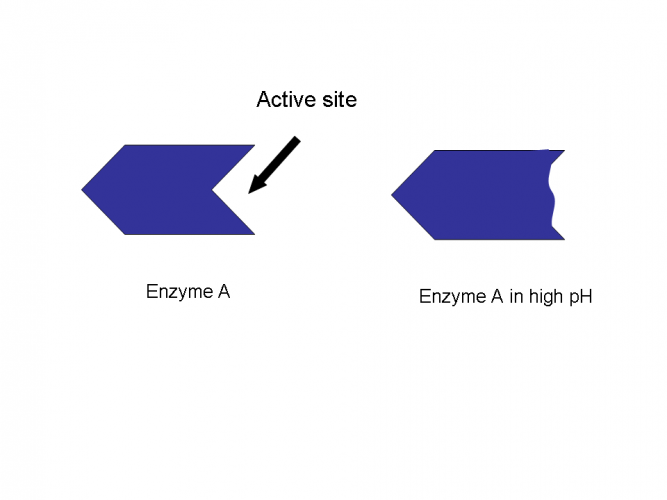
The pH of enzymes surroundings can affect the way the enzymes active site is folded. The active site bonds can break by the changes in pH. This will mean it can become denatured.
Different enzymes work best in different pH's. We say they have an optimum pH. Many enzymes work best in neutral pH (pH 7), whereas some work in acidic conditions while others work better in alkaline conditions. This is often why keen gardeners and farmers will test the soil's pH before growing plants. If the pH is unsuitable then plants will not grow.
The concentration of substrate particles is another factor that affects how quickly an enzyme will catalyse it. The more substrates there are the quicker the rate of reaction will be. After a certain point though, no further reactions can happen. That's because once the enzyme's active site is in constant use and becomes saturated, no more substrates will fit in, even if there's plenty of substrates.
In the following activity, you will determine optimum conditions needed for enzyme function.







