What is photosynthesis and why is it so important to us? The energy from all the food we eat can be traced back to the Sun. This is because plants absorb the Sun's energy and transfer it into glucose (food) during photosynthesis. Oh and photosynthesis also produces oxygen gas, which we need for us to be able to live.
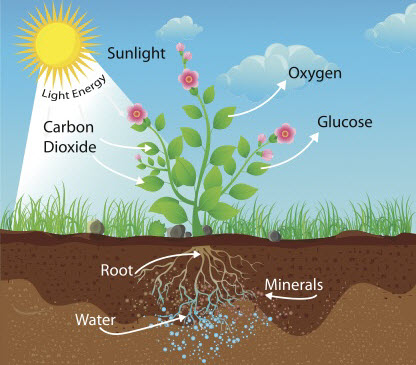
In summary, plants take in carbon dioxide gas and water, and then produce glucose and oxygen gas during photosynthesis.
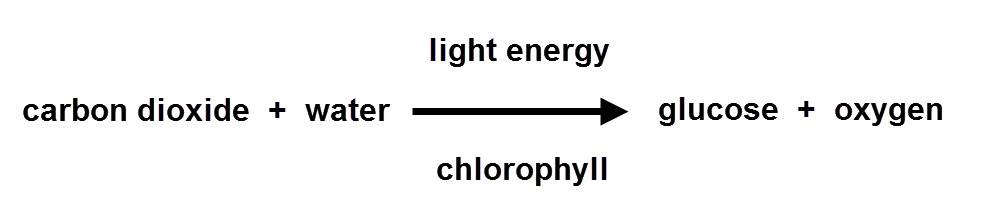
You may have already guessed that the rate (speed) of this reaction depends on the amount of light energy available - photosynthesis will happen a lot faster on a bright, sunny day than on a dull, cloudy day. By actually measuring the volume of oxygen gas produced by a plant, you can work out the rate at which photosynthesis is happening. The more oxygen given off, the higher (or faster) the rate of photosynthesis.
In this worksheet, you will learn to explore how changing the light intensity affects the rate of photosynthesis. You will also discover some of the problems that may arise while collecting data for this experiment.
Aquatic plants are used for this experiment, usually Canadian pondweed. Pondweed will photosynthesise in water - it is put in fish tanks to make sure there is enough oxygen for the fish. If you look closely, you will see oxygen bubbles being produced from the cut end of the stem and this will tell you how quickly photosynthesis is happening.

1. Cut a piece of Canadian pondweed and place this in a beaker of water.
2. Place an inverted (upside down) funnel over the pondweed.
3. Next, fill a measuring cylinder with water and carefully place it over the tip of the funnel.
4. Position a lamp 100 cm away from the beaker (use a ruler to measure this).
5. Start a stopwatch and count the number of bubbles produced by the pondweed in three minutes.
6. After this three minutes is up, look at the measuring cylinder and note down the volume of gas that has been collected.
7. Very carefully, remove the measuring cylinder and refill with water.
8. Repeat this experiment at distances of 80 cm, 60 cm, 40 cm and 20 cm from the beaker. Make sure that you record the number of bubbles and volume of gas collected in three minutes for each distance.

As the distance between the lamp and the pondweed is reduced, the light intensity increases, and your results should show that the rate of photosynthesis increases too. This means that you should be able to count more bubbles and collect a higher volume of gas when the lamp is closer to the pondweed.
Unfortunately, there are a few problems you might come across with this experiment but there are also ways to fix these:
The pondweed might not start the process of photosynthesis straightaway, as it needs time to adjust to the new light intensity. This could be a problem if you are only relying on the counting bubbles method. Wait a couple of minutes before you start the timer or begin counting the bubbles in order to solve this.
Heat given off by the lamp could raise the temperature of the water - temperature is another factor, as well as light intensity, that affects the rate of photosynthesis. This could artificially increase the rate of photosynthesis meaning that the experiment would not be as accurate. It is best to use a lamp with an LED bulb, as this gives off hardly any heat.
The bubbles produced by the pondweed are small and sometimes come out too quickly to be counted accurately. That is why it is a good idea to also collect the volume of oxygen at each light intensity.
Changes in the carbon dioxide concentration may also interfere with your experiment, as this also affects the rate of photosynthesis. You can overcome this by adding sodium hydrogen carbonate to the water in the beaker, as this will keep the level of carbon dioxide constant (the same).
Good scientists will always repeat an experiment at least three times and take the mean average (adding up each value and then dividing the total by the number of values there are) as this gives more accurate results. For example at 80 cm, you may have counted 22 bubbles, 17 bubbles and then 24 bubbles. The mean number of bubbles would be 21 bubbles.
Repeating an experiment will also stop anomalous results (caused by random errors) interfering with your results. Examples of these could be that at one distance you became distracted and lost count of the bubbles, or you talked to your friend and stopped the stopwatch at three minutes 20 seconds instead of three minutes. Errors can also be made by not filling the measuring cylinder completely with water by mistake, or by accidentally changing the angle of the light bulb. If you find an anomalous (an odd) result at one distance, you can exclude that result and then find the mean average. For example at 40 cm, you might have collected 30 cm3, 82 cm3 and then 33 cm3. The second reading is clearly at odds with the other ones, so it is best to ignore that reading and find the mean average of the remaining results for that distance, this would be 31.5 cm3.
Now let's move on to some questions.








