Luke wanted to find out whether the amount of sugar he could dissolve in water could be changed by different factors.
First, he tried to change how much he stirred the sugar and water mixture.

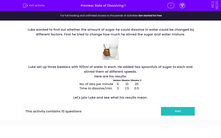
Luke set up three beakers with 100 ml of water in each.
He added two spoonfuls of sugar to each and stirred them at different speeds.
Here are his results:
| Beaker 1 | Beaker 2 | Beaker 3 | |
|---|---|---|---|
| Number of stirs per minute | 5 | 10 | 20 |
| Time to dissolve/min | 3 | 1.5 | 0.5 |
Let's join Luke and see what his results mean.