Everything around you now can be classified as being either a solid, a liquid or a gas. These are the three states of matter. Solids we can see really easily - the chair you're sitting on, the book on your desk, your computer even! Liquids, well we drink some of those - milk, tea, juice - but maybe there's some rain tapping on your window right now - that's a liquid too! Gases are more tricky to spot but they are all around you in the air - oxygen, nitrogen, a bit of carbon dioxide, even water vapour.
Solids, liquids and gases are all made up of tiny particles and the way these particles behave gives the states of matter their characteristic properties.
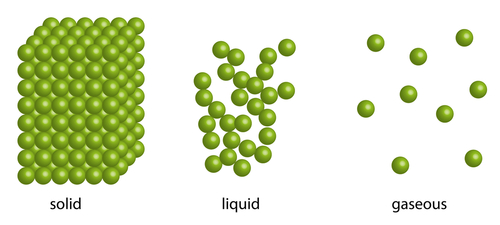
In a solid, the tiny particles that make up the solid are very close together in a neat regular arrangement, all touching each other and moving only by vibrating on the spot. They cannot move much at all, which explains why solids have a fixed shape.

The particles in a liquid are arranged in a more random way. They are close together, most of them touching each other, but there are some small gaps. The particles also have more energy than in solids. So the particles can move around a little, over each other, allowing liquids to flow and be poured. This is the reason why liquids take the shape of the container they are in.

The particles in a gas are spaced very far apart, move around very fast and they are randomly arranged. This is because the particles have lots of energy. Because of this, gases can be easily compressed, or squashed - they have no fixed shape or volume.

Can you use your knowledge of the three states of matter to answer some questions? Great, here we go.......
.jpg)








