Density is an example of a compound measure, which is one that involves more than one unit of measurement.
Density is a measure of mass (e.g. kg, g) in relation to unit volume (e.g. m3, cm3,) and can be measured in kg/m3, g/cm3, etc.
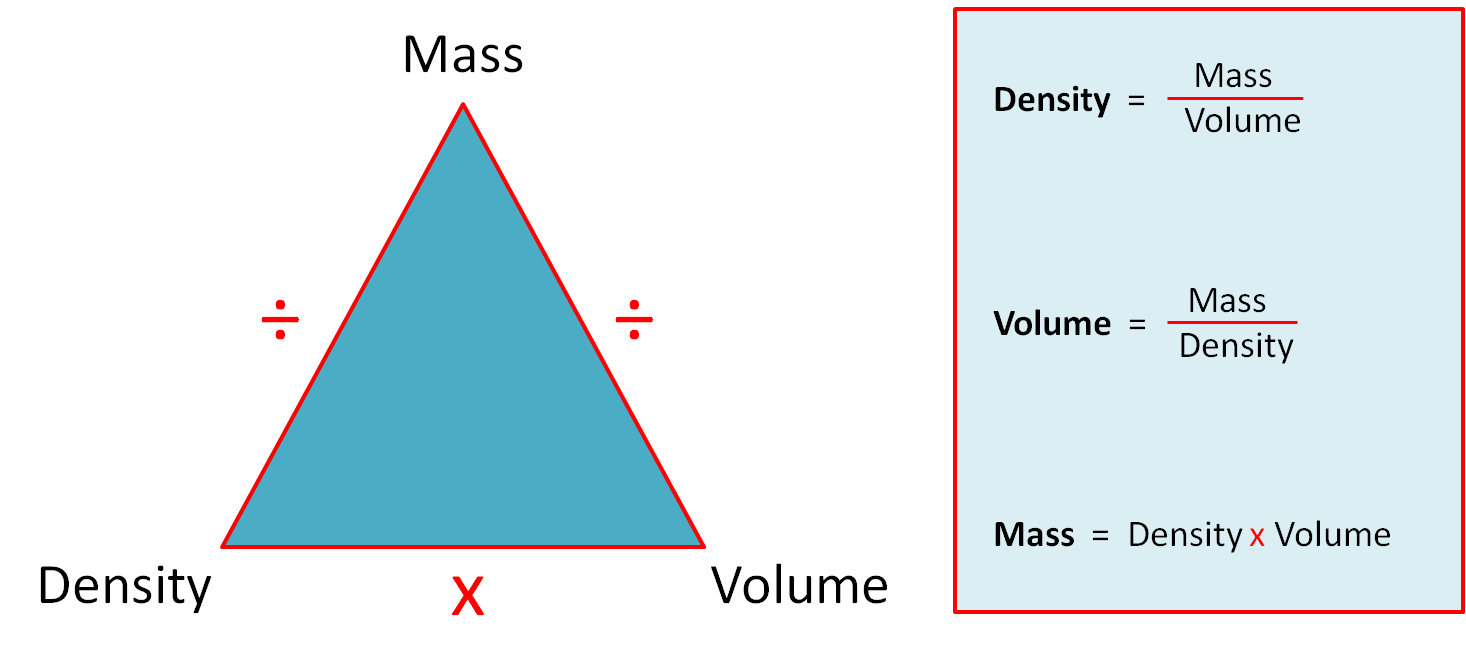
You should know the following formulae and may find the triangle helpful for remembering the three mass, density and volume formulae.
Example 1
A large block of oak has a mass of 185 kg and a volume of 0.25 m3.
Find its density in kg/m3.
Answer
Density = Mass ÷ Volume = 185 ÷ 0.25 = 740 kg/m3.
Example 2
Aluminium has a density of 2.712 g/cm3. Find the mass of a piece of aluminium with a volume of 12 cm3.
Give your answer to 3 sig. figs.
Answer
Mass = Density x Volume
Mass = 2.712 x 12 = 32.5 g
Now let's apply these formulae by tackling some questions together!








