Remember those tiny little things called atoms? Those things that make up everything around us, and even us?! Have you ever wondered, if everything's made up of the same thing, then why isn't everything identical?
.jpg)
Well, there are 118 different types of atom, and that's because there are 118 different elements. Atoms can join up with other types of atoms to form different substances. There are so many different combinations that atoms of these elements can form, and that's why we have such variation and everything is so different from each other. Amazing, hey!

When atoms join together with other atoms of a different type, a compound is formed. Compounds contain different types of atoms chemically bonded together. This means that the atoms are bonded together really tightly which makes separating them very difficult. In order for a compound to be separated, a chemical reaction must take place.
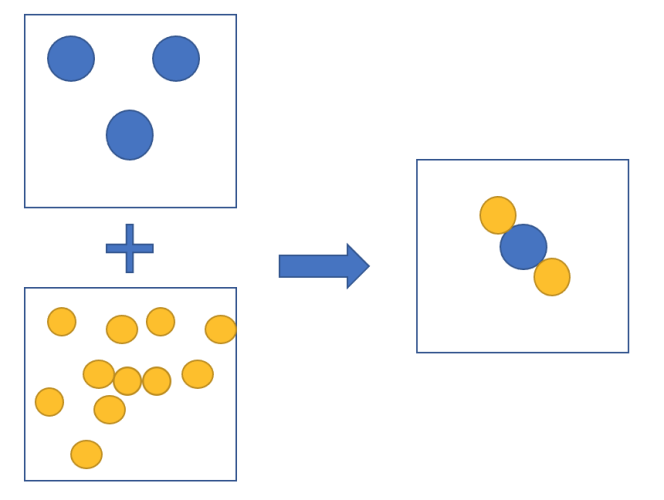
Take a look at the picture above. It shows two elements (one blue, one yellow) joining together to make a compound.

The molecule above is a sulfur dioxide molecule - it is a compound as it is made from two different types of atoms. That means it is made from two different elements. We know they are different types of atoms as they are different colours and sizes. It is made from two atoms of an element called oxygen and one atom of an element called sulfur.
The properties of a compound are often very different to the properties of the elements it is made of. Let's take salt as an example. Common salt that we use in our food is called sodium chloride - it is a compound made from the elements sodium and chlorine. We can't safely eat sodium or chlorine (we'd die if we did!) but when they are joined in a compound, their properties change and we can safely eat it!

By the way, a compound is very different to a mixture. A mixture is a substance consisting of two or more substances, but they are not chemically bonded together. So separating a mixture can be done easily.
Think you've got the idea about compounds now? Lets have a look at some questions together......








