Energy is all around us: we use it, we make it and it is never created or destroyed, but changes forms.
We take in chemical energy from the food we eat, or actually, we use the food we eat to gain energy for us to use in a process called respiration.
Energy in food is often measured in calories (cal), and you may know people who count how many calories they eat each day. In reality, the calories that people count are actually kilocalories (kcal). One kcal contains one thousand cal. However, it is more correct to measure energy of any kind in standard units called joules (J) and to convert calories into joules the number is multiplied by 4.2 (see below).
The amount of energy people need from food each day varies a lot according to age, lifestyle, pregnancy, general health, gender and other possible factors. For an adult, approximately 8000-10,000 KJ (or 2000-2200 calories) are the required daily energy intake.



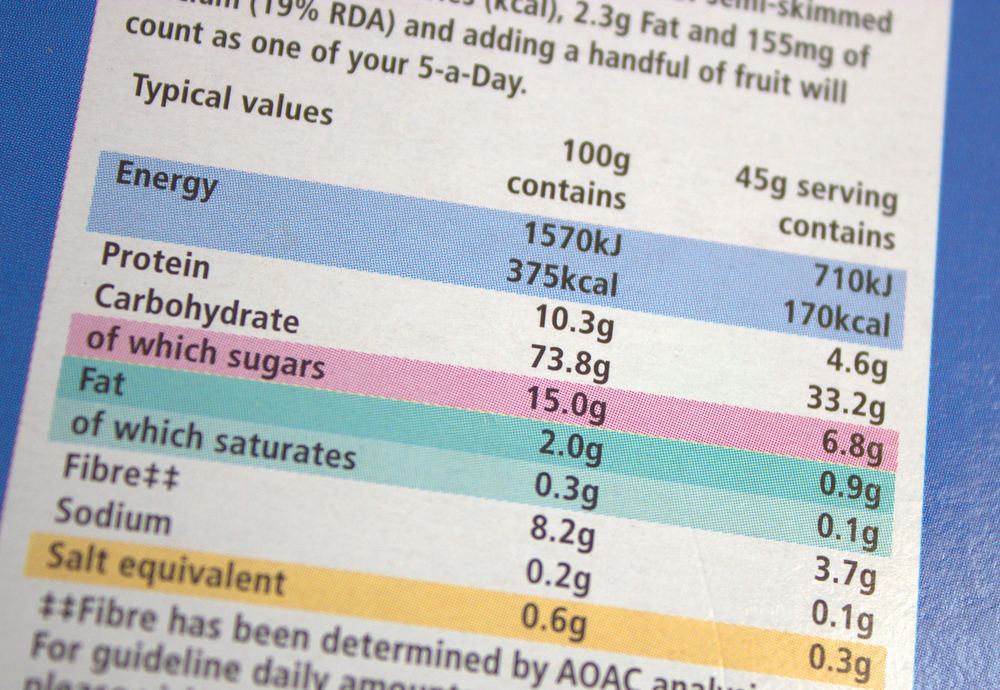
This is a typical content label on a food package. It breaks down the product into its components - for example protein, carbohydrate and fat. The energy ratings are also included, both in J/kJ and cal/kcal.
It is possible to determine the amount of energy in different types of food in a simple experiment. You need different types of food, e.g. crisps and bread, cut up in small pieces. You literally have to burn them under a boiling tube with a certain amount of water and measure how much the temperature rises. The more the temperature of the water increases, the more energy has been released from that type of food.
Be aware that this is an experiment that you may do at school, but you should not try it at home by yourself. The energy released by each food type is calculated using the following formula:
|
energy released from food per gram in Joules |
|
4.2 is a constant number (this means it never changes) and is related to the capacity of water to have its temperature raised.
You may have noticed that, although it was mentioned earlier that calories are used as a unit for energy in foods, in physics energy is measured in joules (J). Protein energy ranges from 2.44 kcal/g to 4.36 kcal/g. Fat has 8.37 kcal/g to 9.02 kcal/g and carbohydrates have 2.70 kcal/g to 4.16 kcal/g.
Now try this activity to see how you manage these energy calculations concerning food.








