When a small piece of potassium is dropped into a trough of water, the result is pretty spectacular. The potassium bursts into flames - purple flames in fact!
.png)
The word equation is:
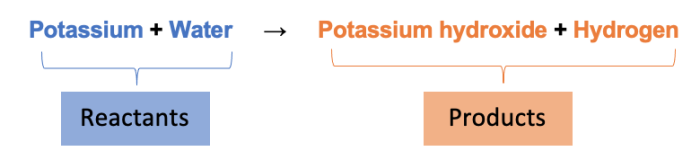
The mass of the two substances before (the reactants) the arrow is always the same as the substances produced (the products)! This is called the Law of Conservation of Mass.
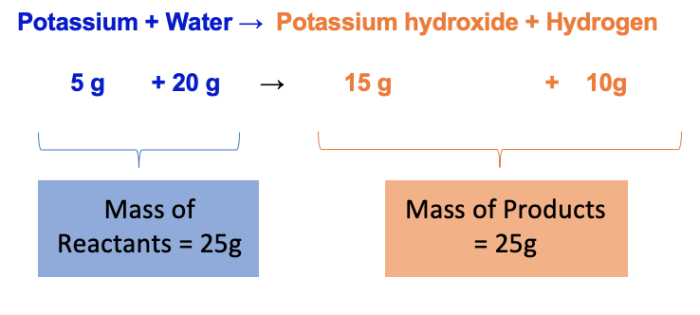
If we knew the mass of the reactants (the mass of potassium and water), and the mass of the potassium hydroxide (which is one of the products), we could work out how much hydrogen gas has also been produced. It's just basically a little maths equation.
Potassium + Water → Potassium hydroxide + Hydrogen
5 g + 20 g → 15 g + ?
The mass of hydrogen produced would be 25 g - 15 g = 10 g
Let's look at some examples together - you might want to have a calculator to hand for some of these questions.









